Why is ANKTIVA® Right for You?

ANKTIVA is an Immunotherapy that Activates Your Body’s Cancer Fighting Cells
ANKTIVA is the first U.S. FDA-approved immunotherapy that activates a type of cell called a natural killer (NK) cell, part of the body’s natural immune system, to attack and kill non-muscle invasive bladder cancer (NMIBC) cells.1 ANKTIVA is a treatment for use in combination with a standard treatment for NMIBC, bacillus Calmette-Guérin (BCG), for people with NMIBC for whom BCG alone was not effective or in whom NMIBC returned after initial successful treatment. These people have what is termed BCG-unresponsive NMIBC.
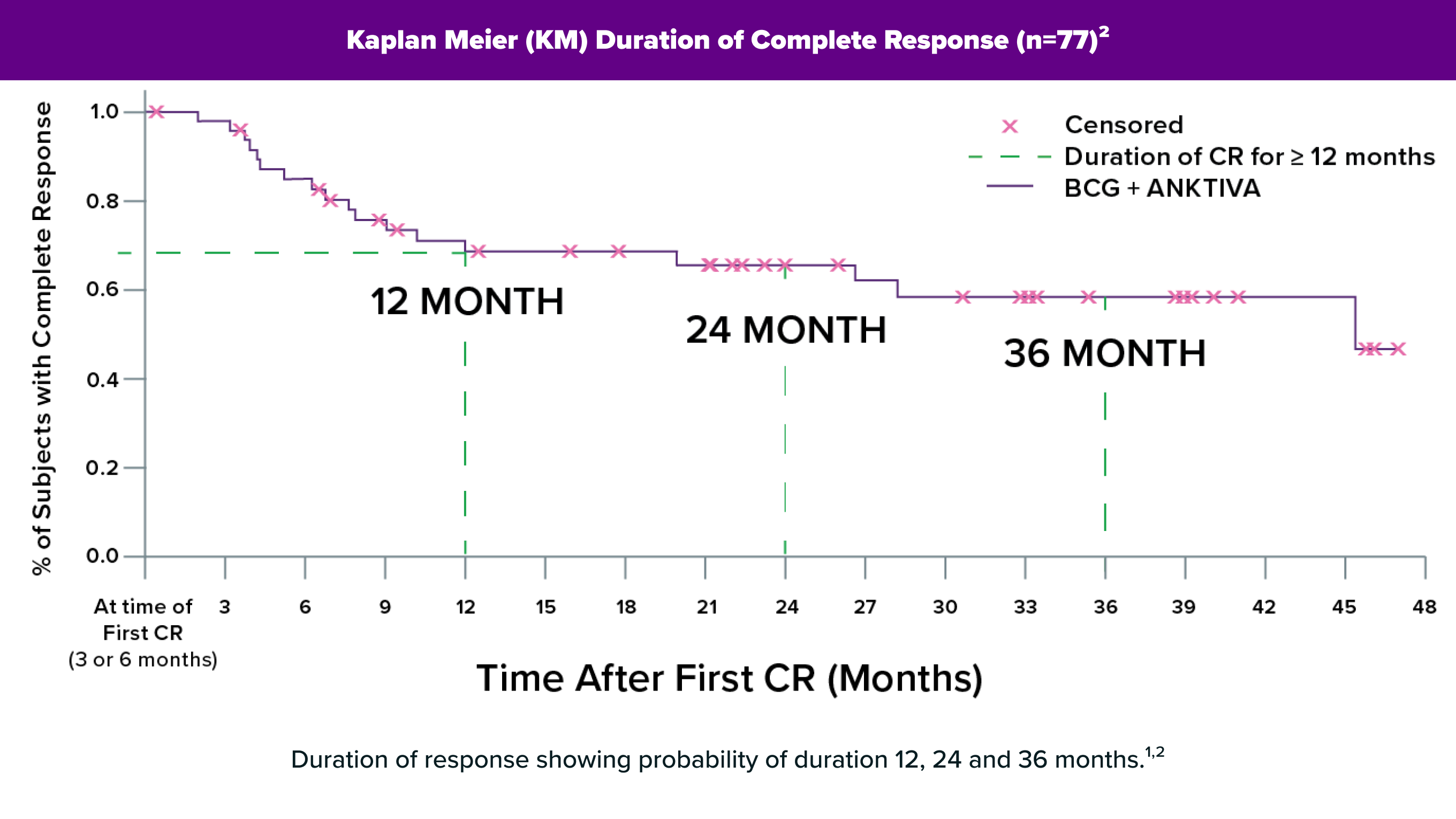
Up to47+ MonthsDuration of Response158% (n=28) ≥12 months; 40% (n=19) ≥24 months; range: 0.0–47.0+ months. + Denotes an ongoing response.
Who Should Consider ANKTIVA?
ANKTIVA is a treatment for adults who have all the following:
- High-risk non-muscle invasive bladder cancer
- NMIBC that did not or is no longer responding to BCG therapy alone (BCG-unresponsive)
- NMIBC that has not grown beyond the inner lining of the bladder to the muscle underneath (called carcinoma in situ or CIS disease)
- NMIBC with or without small finger-like tumor growths that project away from the bladder wall into the bladder (papillary disease)
If You Have Been Diagnosed with BCG-Unresponsive NMIBC CIS Disease and are Interested in ANKTIVA, Here is What You Need to Know:
What ANKTIVA is
- ANKTIVA is an immunotherapy that activates your body’s cancer fighting cells
- ANKTIVA is used in combination with BCG and is administered directly into your bladder via a catheter
- ANKTIVA is the first treatment of its type approved to treat BCG-unresponsive NMIBC
- ANKTIVA can be administered in a clinical setting and allows you to remain in the care of your urologist
What ANKTIVA is not
- ANKTIVA is not a chemotherapy or gene therapy
- ANKTIVA is not administered via an IV and does not circulate throughout your body. It only fights cancer in the bladder. This may reduce side effects
- ANKTIVA is not a chemical, it is a biologic that is similar to molecules produced naturally by your body
- ANKTIVA does not require you to change providers for treatment
Patient Stories
Read about patients who participated in the clinical trials for ANKTIVA
Justin’s Journey
An avid outdoorsman, Justin faced a challenging diagnosis of non-muscle invasive bladder cancer. After struggling with the side effects of his Bacillus Calmette-Guérin (BCG) treatment, he found renewed hope though his doctors' introduction to an immunotherapy for those who experienced a recurrence of NMIBC.
Wayne’s Journey
Wayne has lived a rich life with his wife, two daughters, and three grandchildren. He describes himself as someone who “just likes people.” When he was first told, “you might have a tumor in your bladder,” he became very concerned, especially when he learned his first treatment with BCG wasn’t working. Wayne feels lucky that a friend told him about a clinical trial for patients like himself who were not cured by BCG alone.
ANKTIVA is a Novel Treatment for NMIBC CIS Patients Unresponsive to BCG
For more information, please call 1-877-ANKTIVA