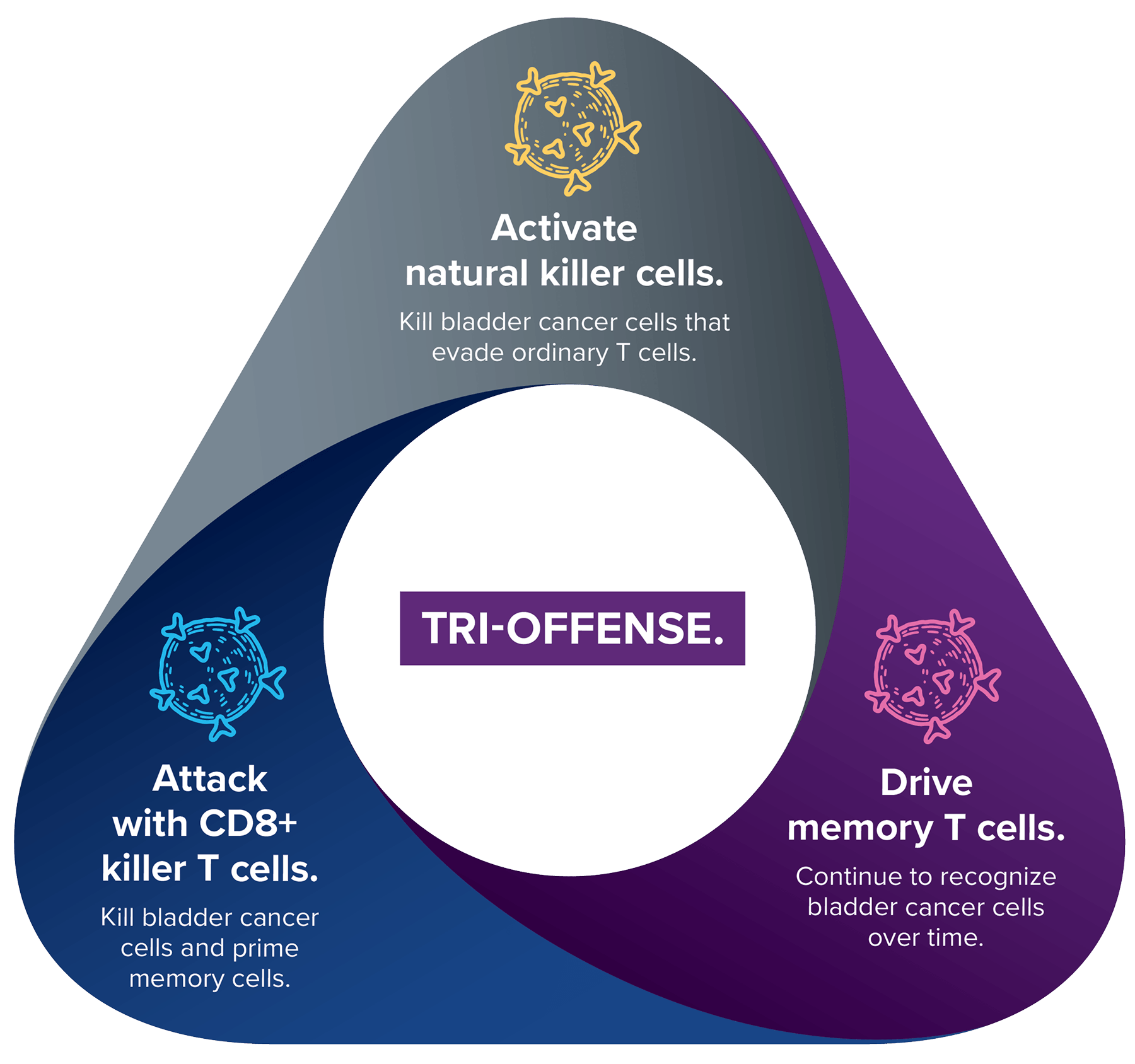
The Tri-Offense: Activating Your Immune Response
In the pivotal clinical trial, treatment of participants with BCG-unresponsive NMIBC with CIS with or without papillary disease with Anktiva plus BCG was found to have complete and durable responses.1
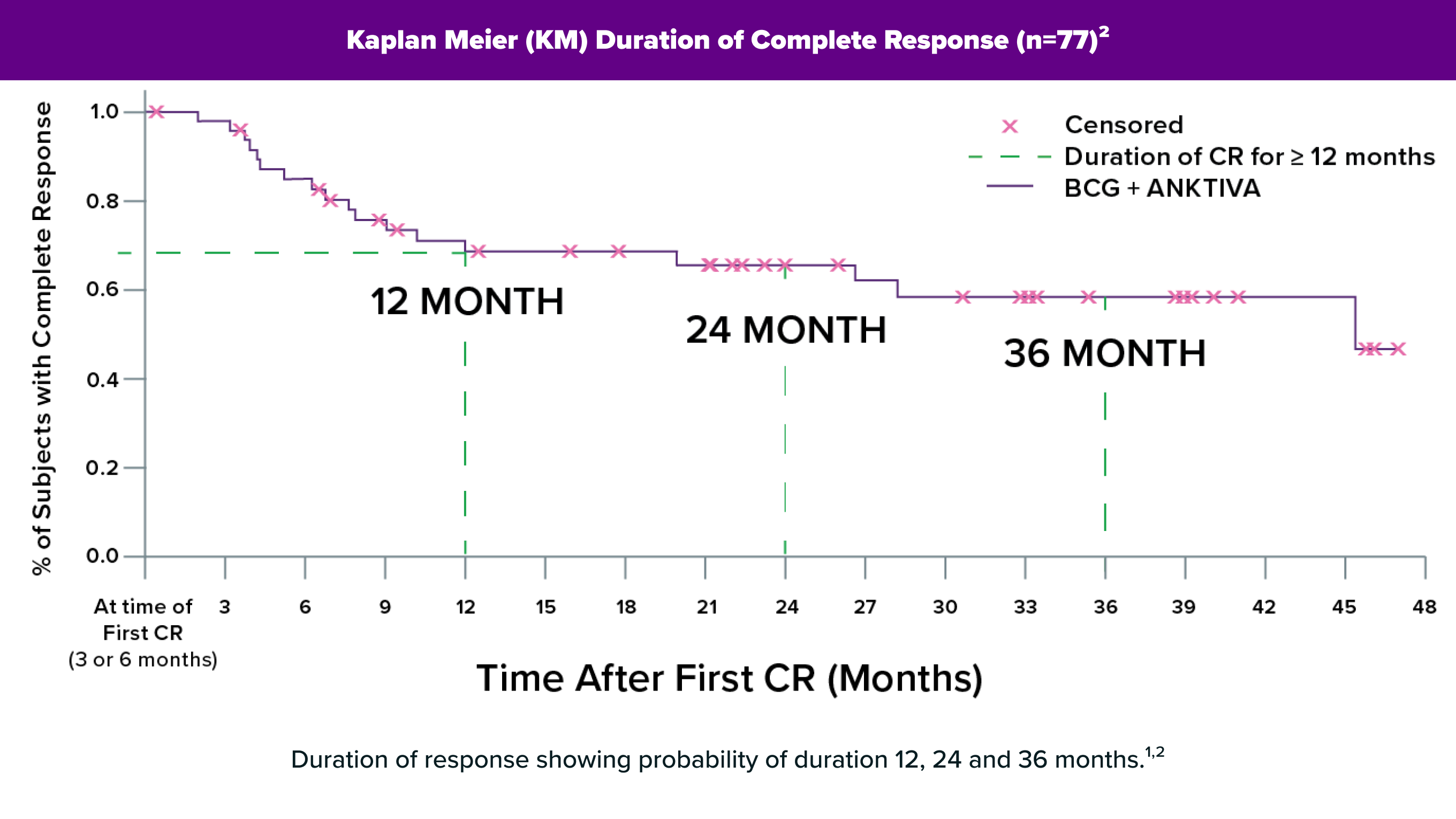
QUILT-3.032 Study

58% (n=28) ≥12 months; 40% (n=19) ≥24 months; range: 0.0–47.0+ months.1
+ Denotes an ongoing response.
Full Enrollment Follow-up
Some patients remained NMIBC-free for over 4 years.2
+ Denotes an ongoing response.
QUILT-3.032 is a multicenter, single-arm study in adults with BCG-unresponsive high-risk NMIBC with CIS ± Ta/T1 papillary disease. Efficacy was evaluated in 77 adults (label), with updated follow-up in 100 patients. Patients received nogapendekin alfa inbakicept-pmln 400 mcg plus BCG weekly for 6 weeks, with re-induction permitted at month 3. The primary endpoint was complete response.1,2
References: 1. Anktiva (nogapendekin alfa inbakicept-pmln) prescribing information. ImmunityBio, 2024. 2. Chang, S. (2025, April 26-29). An Update on QUILT-3.032: Durable Complete Responses to NAI (ANKTIVA) Plus BCG Therapy in BCG-Unresponsive CIS With or Without Ta/T1 Papillary Disease and in Papillary Disease without CIS. [Conference Presentation]. AUA2025, Las Vegas, Nevada, United States.
Adverse Reactions Occurring in ≥15% of Patients in Cohort A in QUILT-3.032
| Adverse Reaction | Anktiva with BCG1 (n=88) | |
|---|---|---|
| All Grades, % | Grades 3 or 4, % | |
| Dysuria | 32 | 0 |
| Hematuriaa | 32 | 3.4 |
| Urinary Frequency | 27 | 0 |
| Micturition Urgencya | 25 | 0 |
| Urinary Tract Infectiona | 24 | 2.3 |
| Musculoskeletal Paina | 17 | 2.3 |
| Chills | 15 | 0 |
| Pyrexia | 15 | 0 |
Clinically relevant adverse reactions in <15% of patients who received Anktiva with BCG included fatigue (14%), nausea (14%), bladder irritation (11%), diarrhea (9%), and nocturia (7%).
Efficacy Results in QUILT-3.032
| Anktiva with BCG1 (n=77) | |
|---|---|
| Complete Response Rate (95% CI) | 62% (51, 73) |
| Duration of Responsea | |
| Range in months | 0.0, 47.0+ |
| % (n) with duration ≥ 12 months | 58% (28) |
| % (n) with duration ≥ 24 months | 40% (19) |
a. Based on 48 patients that achieved a complete response at any time; reflects period from the time complete response was achieved

Getting Started With Anktiva
See how Anktiva works with BCG in the Tri-offense and what to expect before and after treatment
Patient Stories
Patients Like You
Read about patients who participated in the clinical trials for Anktiva
Justin’s Journey
An avid outdoorsman, Justin faced a challenging diagnosis of non-muscle invasive bladder cancer. After struggling with the side effects of his Bacillus Calmette-Guérin (BCG) treatment, he found renewed hope though his doctors' introduction to an immunotherapy for those who experienced a recurrence of NMIBC.
Wayne’s Journey
Wayne has lived a rich life with his wife, two daughters, and three grandchildren. He describes himself as someone who “just likes people.” When he was first told, “you might have a tumor in your bladder,” he became very concerned, especially when he learned his first treatment with BCG wasn’t working. Wayne feels lucky that a friend told him about a clinical trial for patients like himself who were not cured by BCG alone.

Anktiva Resources
Learn more about the clinical story of Anktiva
Future Oncology
A plain language review of the published findings on the Phase 1b BCG-naïve and Phase 2/3 BCG-unresponsive studies (QUILT 3.032).
When BCG Alone Isn’t Enough,
Bring On the Tri-Offense With Anktiva.
For more information, please call 1-877-ANKTIVA.