Think ANKTIVA®.
ANKTIVA is the first FDA-approved immunotherapy designed to activate the body’s natural immune system to target and attack BCG-unresponsive non-muscle invasive bladder CIS (NMIBC CIS), potentially leading to a long duration of complete response up to 47+ months1.

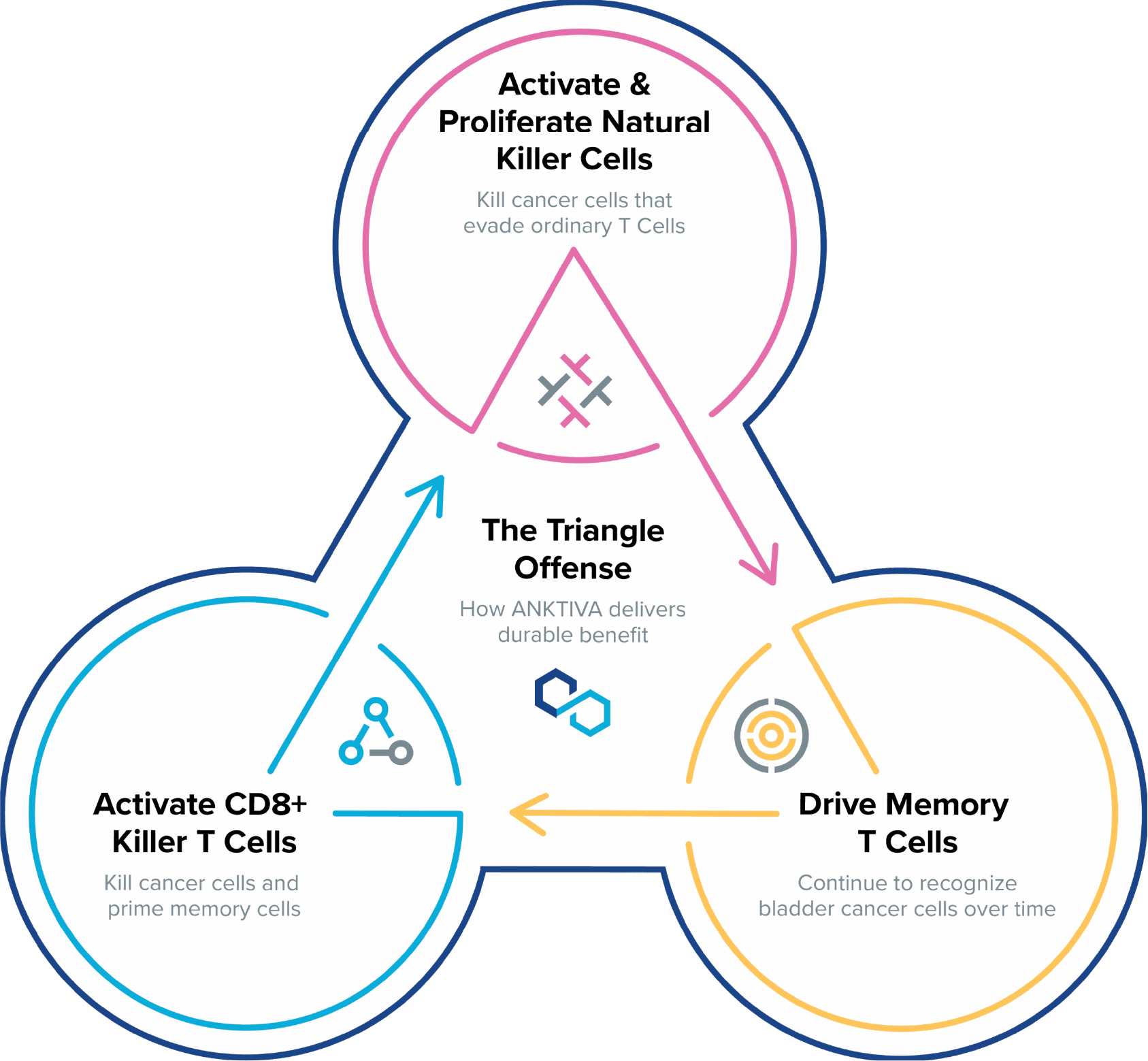
ANKTIVA is a Potent Immunostimulant that Unlocks Anti-tumor Activity and Unleashes Durable, Complete Responses.
ANKTIVA uniquely stimulates the body’s natural immune system by proliferating and activating natural killer cells, as well as cytotoxic and memory T cells to drive long-term immune memory and durable responses.

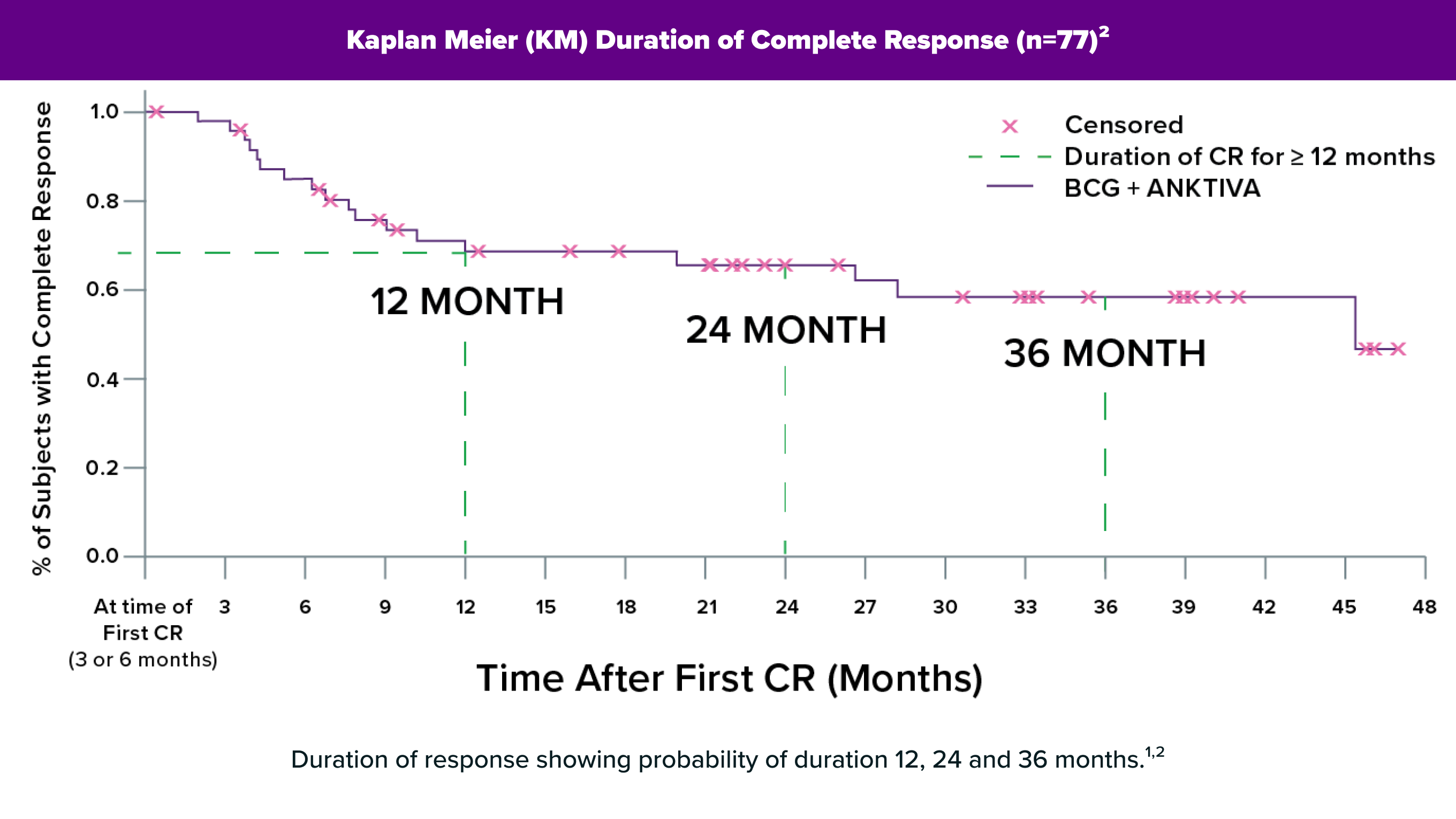
Duration of Response
Up to47+ Months58% (n=28) ≥12 months; 40% (n=19) ≥24 months; range: 0.0–47.0+ months1+ Denotes an ongoing response.
Duration of ResponseUp to53 MonthsSome patients remained NMIBC-free for over 4 years2
1. Anktiva (nogapendekin alfa inbakicept-pmln) prescribing information. ImmunityBio, 2024. 2. Chang, S. (2025, April 26-29). An Update on QUILT-3.032: Durable Complete Responses to NAI (ANKTIVA) Plus BCG Therapy in BCG-Unresponsive CIS With or Without Ta/T1 Papillary Disease and in Papillary Disease without CIS. [Conference Presentation]. AUA2025, Las Vegas, Nevada, United States.
Adverse Reactions Occurring in ≥15% of Patients in Cohort A IN QUILT-3.032
| Adverse Reaction | ANKTIVA with BCG1 (n=88) | |
|---|---|---|
| All Grades % | Grades 3 or 4 | |
| Dysuria | 32 | 0 |
| Hematuria1 | 32 | 3.4 |
| Urinary Frequency | 27 | 0 |
| Micturition Urgency1 | 25 | 0 |
| Urinary Tract Infection1 | 24 | 2.3 |
| Musculoskeletal Pain1 | 17 | 2.3 |
| Chills | 15 | 0 |
| Pyrexia | 15 | 0 |
Clinically relevant adverse reactions in <15% of patients who received ANKTIVA with BCG included fatigue (14%), nausea (14%), bladder irritation (11%), diarrhea (9%), and nocturia (7%).
Efficacy Results in QUILT-3.032
| ANKTIVA with BCG1 (n=77) | |
|---|---|
| Complete Response Rate (95% CI) | 62% (51, 73) |
| Duration of Responsea | |
| Range in months | 0.0, 47.0+ |
| % (n) with duration ≥ 12 months | 58% (28) |
| % (n) with duration ≥ 24 months | 40% (19) |
a. Based on 48 patients that achieved a complete response at any time; reflects period from the time complete response was achieved
Patient Stories
Read about patients who participated in the clinical trials for ANKTIVA
Justin’s Journey
An avid outdoorsman, Justin faced a challenging diagnosis of non-muscle invasive bladder cancer. After struggling with the side effects of his Bacillus Calmette-Guérin (BCG) treatment, he found renewed hope though his doctors' introduction to an immunotherapy for those who experienced a recurrence of NMIBC.
Wayne’s Journey
Wayne has lived a rich life with his wife, two daughters, and three grandchildren. He describes himself as someone who “just likes people.” When he was first told, “you might have a tumor in your bladder,” he became very concerned, especially when he learned his first treatment with BCG wasn’t working. Wayne feels lucky that a friend told him about a clinical trial for patients like himself who were not cured by BCG alone.
ANKTIVA Resources
Information for ANKTIVA
ANKTIVA: How to Order
A step-by-step guide on how to order ANKTIVA, including product codes

Physicians' Journey
Listen to the physicians' journey with their patients using ANKTIVA.
ANKTIVA is a Novel Treatment for NMIBC CIS Patients Unresponsive to BCG
For more information, please call 1-877-ANKTIVA